We had a very serious problem with our resistance to antibiotics. Now we are closer to solving it
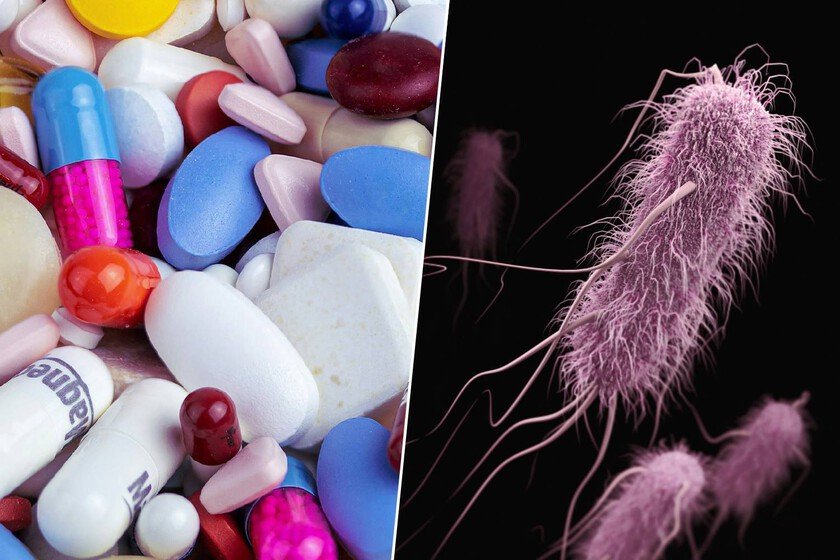
One of the great threats that humanity faces today is without a doubt the antibiotic resistancewhich leads to emergence of bacteria that are resistant to all pharmacological weapons that we have. This forces science to have to look for new antibiotics and new ways to ‘attack’ a bacteria. And at the moment it seems that we are approaching this great milestone with a new antibiotic that was hidden in plain sight. The problem. Having bacteria that you cannot compete against is undoubtedly a death sentence for the person who is unlucky enough to be its host. Something that responds to the mechanisms that these microorganisms have to evolve and develop ‘tactics’ that allow them to escape our antibiotics. A very typical situation in a hospital, especially where a bacteria that has been exposed to a treatment, but has survived, will adapt to that environment. This makes the WHO categorize antimicrobial resistance as “one of the top 10 threats to global public health.” Put another way: we are running out of antibiotics that work, since bacteria are evolving faster than we are discovering new drugs. And this is something that is also magnified by our own fault by taking antibiotics uncontrollably or not complete treatment guidelines appropriately. That is why the discovery just made by a team from the University of Warwick and Monash University is so spectacular: have found a “silver bullet” that had been hidden in plain sight for 50 years. The discovery. Published in it Journal of the American Chemical Societywe are talking about an antibiotic that, in early tests, has been shown to be up to 100 times more powerful than existing drugs against high-priority resistant bacteria, such as feared Staphylococcus aureus methicillin resistant (MRSA). The molecule in question is called pre-methylenemycin C lactone (compound 5), and it has arrived to try to save humanity from this pandemic we are experiencing. But the most surprising thing is where they found it: in the Streptomyces coelicolora soil bacteria that is literally the “model organism” for the production of antibiotics and which has been studied endlessly since the 1950s. That is to say, we had a possible solution before our eyes and we had not realized it until now. This bacteria produces a well-known antibiotic called methylenemycin A that is low potency and is not used clinically. However, scientists decided to investigate not only the final product, but the intermediate steps of its biological “assembly line.” This is where it was seen that it intermediately produced methylenemycin C, which has much more powerful antimicrobial effects. And this is a lesson for science: we are always left with the result of the reactions (that is, the final product). But now what should be done is analyze everything that happens between the first substrate and the final product. Because we are seeing how methylenemycin A was discovered 50 years ago and it was not until now that one of its intermediate products has been a protagonist in this fight. As. To achieve this, the team used genetic engineering. Basically, they “sabotaged” the bacteria’s production chain by creating a mutation that eliminated the gene. mmyE. When this piece is missingthe bacteria could no longer complete the process and began to accumulate the “intermediate steps.” Something similar to when in a production line we remove one of the tapes and an intermediate version of what we were manufacturing begins to accumulate. The tests. When they tested the activity of the new molecule, the results were astonishing. Compound 5 (pre-methylenemycin C lactone) was “one to two orders of magnitude more active” (i.e., 10- to 100-fold) than methylenemycins A and C (the final products). In this way, it was finally possible to see that the result was up to 256 times more powerful than even some drugs. Something that is revolutionary. The great hope. Being powerful is all well and good, but the real battle is against resistance. That is, when the bacteria come into contact with this antibiotic, they can develop a system to get rid of its lethal effect. And this is where there is good news, since after subjecting the bacteria E. faecium At increasing concentrations of the new antibiotic for 28 consecutive days, a standard method for forcing the emergence of resistance, no resistance was detected. A new way to search. Until now, the intermediate products generated in the production of different medicines had been ignored. Now this study puts an end to this custom, since it has become clear that the identification and testing of the intermediate elements of biosynthesis can lead to a great revolution. Now with this new treatment, preclinical tests with animals remain to assess its safety with the aim of subsequently moving on to tests in humans and the evaluation of its side effects. Images | CDC Myriam Zilles In Xataka | AI has opened a chest that had been closed for almost 4 billion years: the salvation of antibiotics