Trapping carbon dioxide emissions and literally turning them into stone seems like an invention straight out of the blue. Futuramawhere in the future everything is recycled. The problem is that this trick of underground alchemy hid a terrifying small print: his exorbitant thirst. To get carbon to mineralize underground, the system needs to swallow absurd amounts of liquid, specifically between 20 and 50 times more water than the mass of CO₂ we are trying to store.
However, a new industrial-scale study published in the magazine Nature just rewritten the rules of the game. An international team, with researchers from Iceland, Saudi Arabia and Italy, has shown in the western Saudi desert that it is possible to petrify CO₂ without wasting a single drop of external fresh water.
Salvation under the sands of Saudi Arabia. As the authors of the research detail, this area is a real challenge: it is full of large facilities that emit a lot of CO₂, such as refineries and desalination plants, but it lacks the underground saline aquifers or sedimentary traps that are traditionally used to inject carbon.
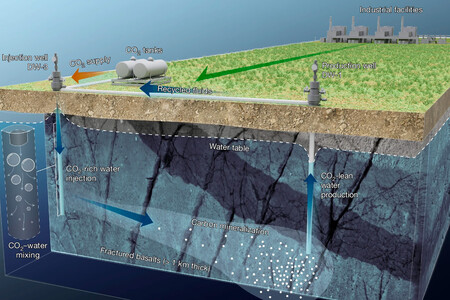
Salvation was under his feet. About 24 kilometers from the Jizan Economic Complex and Refinery, geologists took advantage of an immense bed of highly fractured volcanic rocks (basalts) that have been there for between 21 and 30 million years. There they tested an ingenious system for recirculating subsoil fluids.
The gigantic “soda” trick. To carry out the experiment, the engineers used two main wells, separated by just 130 meters: one functions as a “production” well (extracts water) and the other as an “injection” well. The process is a closed circuit and isolated from the atmosphere so that oxygen does not enter or gas escape. They extract the water that already lives in the depths, circulate it through pipes and, 150 meters underground, inject pure CO₂ into it in the form of bubbles until it completely dissolves.
According to the project scientists, dissolving the gas in water has two brutal chemical and mechanical advantages:
- It gets heavy: CO₂-laden water is denser than regular still water, so it creates a non-buoyant fluid, greatly limiting the risk of the gas migrating to the surface and back into the atmosphere.
- It becomes acidic: This liquid is acidic and greatly accelerates the dissolution of the silicate minerals present in the basaltic rock. As the rock dissolves, it releases metals that provide the cations needed to form stable minerals, such as calcite.

A question of geopolitical survival. The data from this pilot is a resounding success. The team injected 131 tons of CO₂ into the subsoil. After monitoring the area with trackers, they discovered that approximately 70% of all that injected carbon had been mineralized within ten months. Measurements showed that the concentration of dissolved inorganic carbon in the returning water had been reduced by 90% compared to what was initially injected.
Reusing water from the reservoir itself offers substantial advantages. Not only do you forget about bringing external water, but you also reduce the risk of the pressure of fluids underground increasing dangerously. Furthermore, by injecting water that has the same composition as the original underground reserve, the risk of compatibility problems, such as losses of permeability in the reservoir, is reduced.
The current dimension. As we recently analyzed in Xataka In the wake of military escalation in the region, the real Achilles heel of the Arabian Peninsula is not oil, but thirst. Countries like Saudi Arabia depend 70% on their desalination plants to survive. In a scenario where the supply of fresh water is a strategic vulnerability and a matter of biological survival, allocating massive volumes of water to bury emissions was simply unfeasible. Therefore, this advance opens the door for the Middle East – where a large part of global oil production is also concentrated – to be able to use its basalt rocks to store carbon without sacrificing a vital resource.
A providential accident. Sometimes setbacks are the best of tests. In September 2023, the submersible pump in the extraction well broke down. When the technicians brought it to the surface, they found its interior full of rock grains cemented by up to 14% calcite, as well as other minerals such as siderite and ankerite. The isotope analyzes made it clear: these solid cements were formed from the CO₂ injected during the pilot project. The gas had literally petrified in the very bowels of the machine.
An “energy bargain”. As if that were not enough, we must add energy savings. As the research details, injecting CO₂ with this method requires a surface pressure of only 12 to 14 bars. That’s 8 to 16 times less pressure than conventional carbon capture plants require. Basically, CO₂-laden water is drawn into the system driven by gravity. Regarding its future potential, engineers calculate that the underground pores of this particular area (estimated between 24,000 and 43,000 m³) would have enough space to house between 22,000 and 40,000 tons of mineralized CO₂.
Geology dictates: the limit of the stone. Every geological technology has its own physical limits. As experts explain Natureas water, CO₂ and basalt interact, the total volume of solid minerals increases. This means that the pore space is reduced and can end up blocking water flow paths in the long term. To get around this problem, the researchers propose that we may have to resort to fracturing the rock (fracking), an option still little explored in basaltic systems.
What is clear is that this technological innovation is proposed as a great complement to conventional capture systems, not as an exclusive alternative, since in the end it is the geological conditions that rule. But thanks to this pioneering experiment, there is something we can take for granted: the lack of rivers or fresh aquifers is no longer an excuse for not returning our emissions to the subsoil and turning them into stone.

GIPHY App Key not set. Please check settings